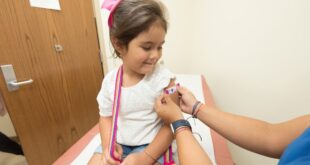
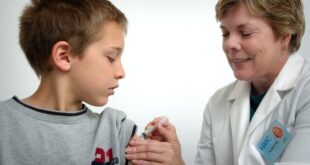
The Centers for Disease Control and Prevention recommended Saturday that children as young as six months old receive Covid-19 vaccines developed by Pfizer-BioNTech and Moderna. The move follows a unanimous vote earlier in the day by an advisory panel of outside experts known as Advisory Committee on Immunization Practices, or …
Read More »FDA Advisers Recommend Pfizer, Moderna COVID Vaccines For Kids Under 5
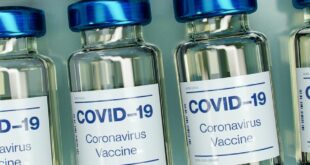
Parents of children under 5 are one step closer to vaccinating their young kids against COVID-19 after the U.S. Food and Drug Administration’s outside vaccine panel recommended emergency use authorizations of two COVID-19 vaccines Wednesday. The 21-person Vaccines and Related Biological Products Advisory Committee voted unanimously to recommend the FDA …
Read More »Under Fire From Congress, FDA Chief Says Baby Formula Shortage Will Improve Soon
by Jennifer Shutt, Georgia Recorder May 19, 2022 WASHINGTON — U.S. Food and Drug Commissioner Robert Califf told Congress Thursday that the infant formula shortage will likely begin to improve in the coming days, though he declined to explain to unhappy lawmakers why his agency didn’t act sooner on a …
Read More »Baby Formula Maker Abbott Reaches FDA Deal To Reopen Plant In Two Weeks
Months after it stopped production and recalled its products, worsening a pandemic formula shortage, baby formula maker Abbott has entered into a consent decree with the U.S. Food and Drug Administration, making way for the pharmaceutical company to reopen its infant formula plant in Michigan, Abbott announced Monday. Once the …
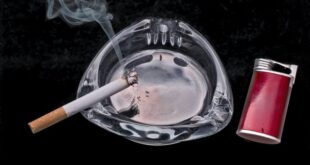
Read More »FDA Moves To Ban Sales Of Menthol Cigarettes, Flavored Cigars
The U.S. Food and Drug Administration (FDA) announced it has begun the process of banning menthol cigarettes and flavored cigars. “Banning menthol — the last allowable flavor — in cigarettes and banning all flavors in cigars will help save lives, particularly among those disproportionately affected by these deadly products. With these …
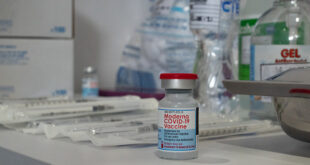
Read More »FDA Authorizes First COVID-19 Breath Test
The Food and Drug Administration (FDA) has granted emergency use authorization to a new COVID-19 test that can detect infections with only a sample of a patient’s breath, using a device that can yield results in less than three minutes. The agency says the InspectIR COVID-19 Breathalyzer will only be available for …
Read More »Biden’s Pick For FDA Commissioner Confirmed By Senate
On Tuesday, Robert Califf was confirmed as the United States Food and Drug Administration (FDA) commissioner in a 50-46 Senate vote in which 6 Republicans supported him, and 5 Democrats voted against his confirmation. Republican Sen. Mike Rounds of South Dakota voted “present,” in what is called a “live pair” …
Read More »Moderna COVID-19 Vaccine Gets Full FDA Approval
On Monday, the Food and Drug Administration (FDA) gave full approval to Moderna‘s COVID-19 vaccine for people 18 years of age and older. The approval makes it the second vaccine to be fully licensed in the United States. The vaccine will be marketed as Spikevax. Moderna’s messenger RNA vaccine has …
Read More »CDC recommends Pfizer’s COVID-19 booster for kids ages 12 to 15
The Centers for Disease Control and Prevention (CDC) has endorsed the Food and Drug Administration (FDA) and the Advisory Committee on Immunization Practices’ (ACIP) recommendations to expand eligibility of Pfizer’s COVID-19 booster doses to kids 12 to 15 years old. CDC now recommends that adolescents aged 12 to 17 years …
Read More »FDA Authorizes Pfizer’s Booster Shot For Children Aged 12-15-Years-Old
On Monday, the Food and Drug Administration (FDA) extended the emergency use authorization for the Pfizer and BioNTech Covid-19 booster shot to include children aged 12 to 15. “Throughout the pandemic, as the virus that causes COVID-19 has continuously evolved, the need for the FDA to quickly adapt has meant …
Read More »FDA authorizes second COVID-19 antiviral pill, Merck’s molnupiravir
One day after the U.S. Food and Drug Administration (FDA) granted emergency use authorization to Pfizer’s COVID-19 antiviral pill, Paxlovid, the agency on Thursday granted the same emergency use authorization to Merck’s antiviral medication, molnupiravir. Both pills are intended only for people who are at high risk for hospitalization and death from severe …
Read More »CDC Expands COVID-19 Booster Recommendations To 16, 17-Year-Olds
16 and 17-year-olds can receive a booster shot for Pfizer’s COVID-19 vaccine after the Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) approved the additional dose Thursday. The Food and Drug Administration gave emergency authorization for 16- and 17-year-olds to receive a third dose of the Pfizer …
Read More »FDA Advisors Vote To Recommend Authorization Of Merck’s Covid Pill
A Food and Drug Administration (FDA) advisory panel on Tuesday narrowly voted to endorse Merck’s COVID-19 pill in a 13-10 vote. If the drug is authorized, this will be the first treatment for the virus Americans can use at home. The panel’s recommendation is not binding, and the Food and Drug …
Read More »All Adults In U.S. Now Eligible For COVID Booster Shots
COVID-19 booster shots are now available to all adults in the United States, after Centers for Disease Control and Prevention (CDC) Director Rochelle Walensky signed off on the recommendations of an agency advisory panel on Friday. “After critical scientific evaluation, today’s unanimous decision carefully considered the current state of the pandemic, the …
Read More »CDC Recommends COVID-19 Vaccine for Children 5 to 11 Years
On Tuesday, CDC Director Dr. Rochelle Walensky endorsed the CDC Advisory Committee on Immunization Practices’ (ACIP) recommendation that children 5 to 11 be vaccinated against COVID-19 with the Pfizer-BioNTech pediatric vaccine. The move clears the way for shots to be administered as soon as tomorrow, though it may be a few days before the vaccine is widely …
Read More » Poli Alert Politics & Civics
Poli Alert Politics & Civics